|
7/21/2023 0 Comments Covid illinois 1b  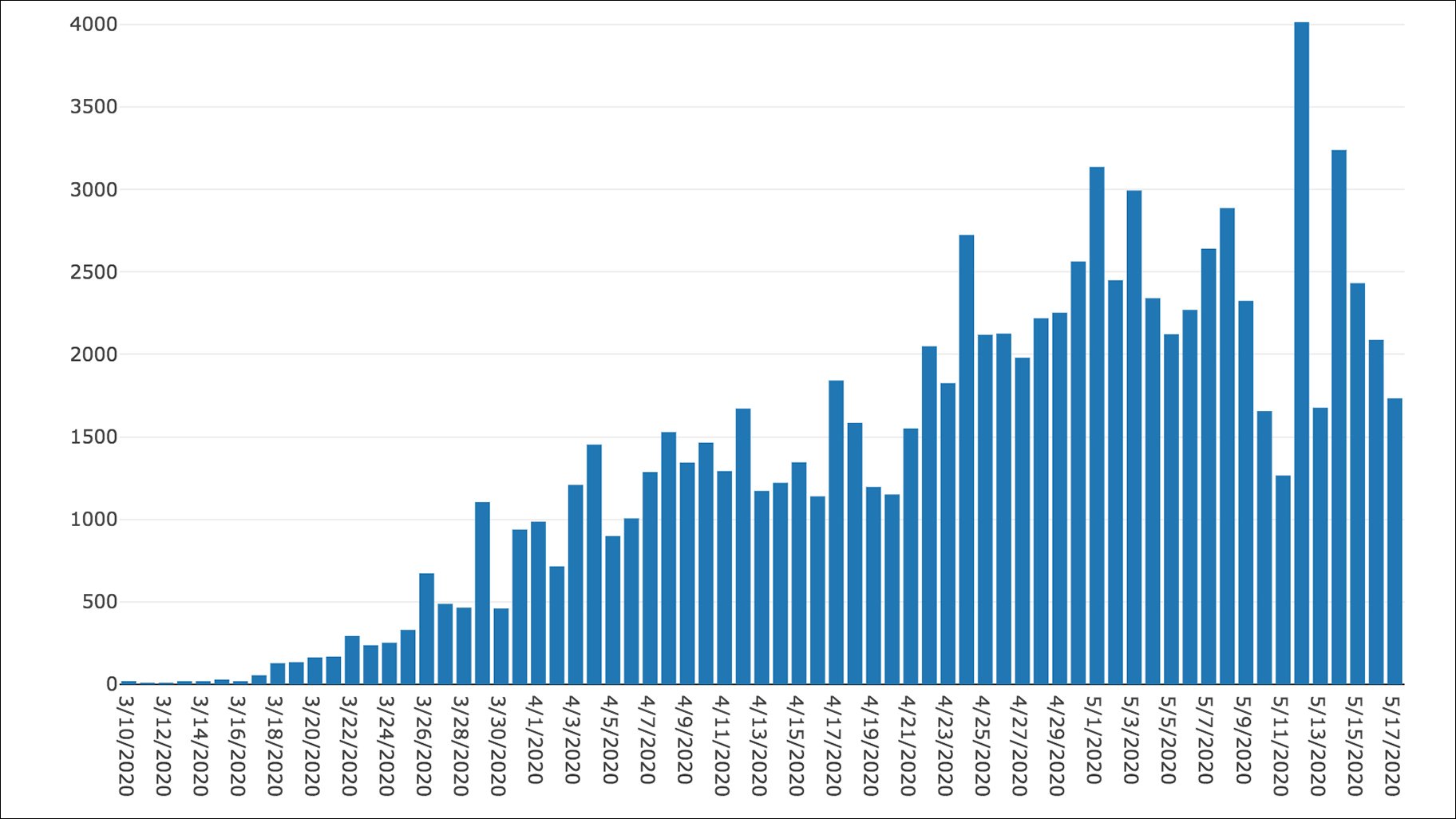
(C) Seroprevalence of autoantibodies in patients with ongoing PASC and individuals after infection without developing PASC. (B) Seroprevalence of autoantibodies over time in COVID-19 patients. Serological profiling of plasma from patients of the discovery cohort with ongoing PASC, after resolved SARS-CoV-2 infection and after resolved PASC (A) Proportion of participants with rheumatic factor (RF), and antinuclear (ANA) and phospholipid autoantibodies (aPL) dependent on COVID-19 history. (J) Post-vaccination status of patients with ongoing PASC. Abp, abdominal pain An, angina Ax, anxiety Ba, body aches Ca, coryza Co, cough Cv, conjunctivitis De, depression Di, dizziness Dy, dyspnea Fa, fatigue Fe, fever Gc, gastrointestinal complaints He, headache Hc, heart complaints Lc, lack of concentration Ls, lymph node swelling Lts, loss of taste/smell Na, nausea Sai, self-reported severity of acute infection SD, sleep disturbance St, sore throat Ti, tinnitus. 
(I) Severity of acute COVID-19 in PASC patients. Bars represent range from smallest to highest value. (H) Age distribution of DigiHero participants with or without PASC shown as box plot extending from the 25th to 75th percentiles. (G) Distribution of PASC duration between female and male study participants with prior COVID-19. (F) Severity of self-reported symptoms in PASC patients. (E) Proportion of PASC patients with mild/moderate or at least one severe symptom. 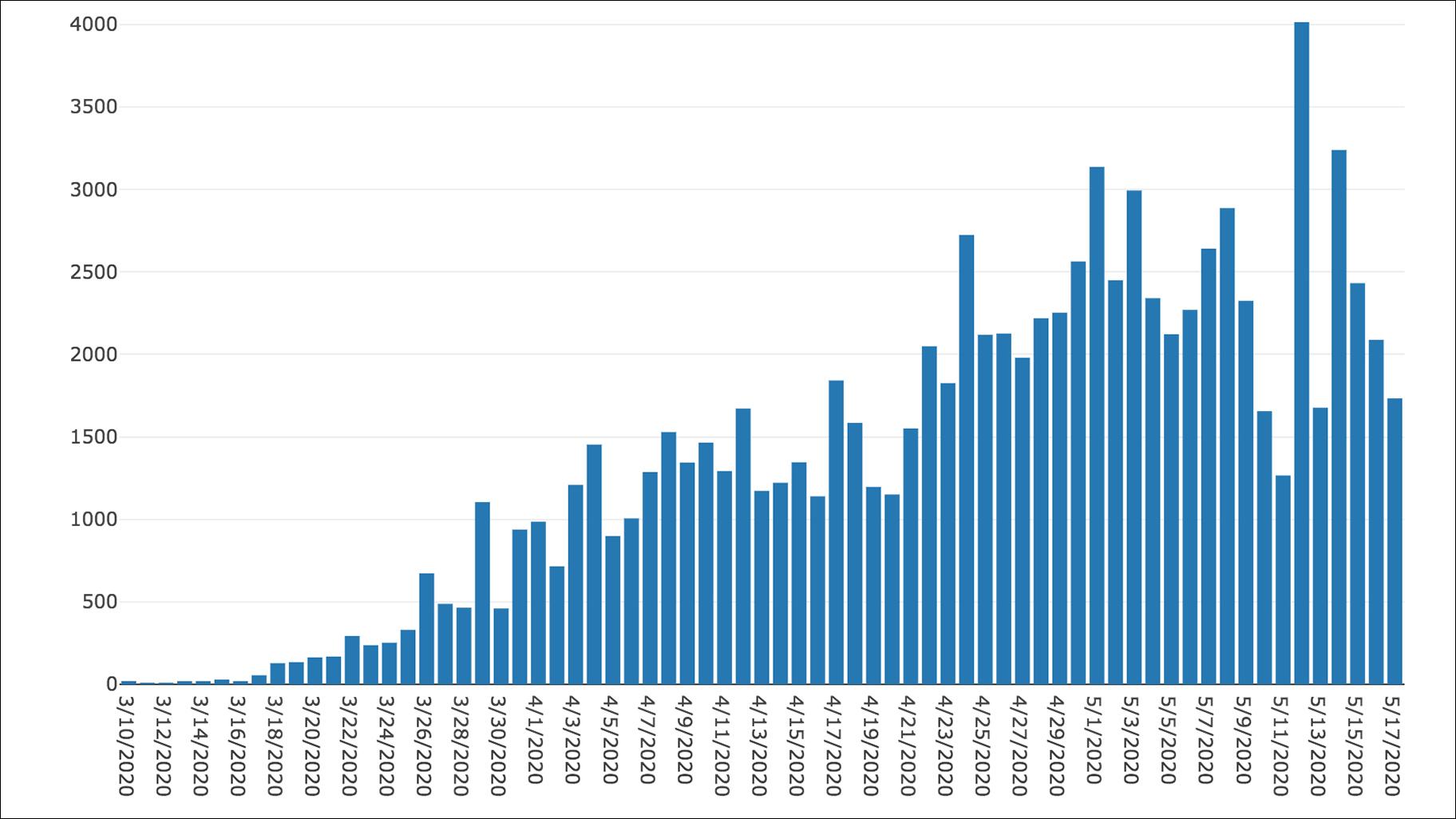
(D) Proportion of DigiHero participants with self-reported PASC including duration of PASC symptoms after infection plus proportion of patients with ongoing symptoms at the time of blood sampling. (C) Plasma titer of antibodies directed against the S1 and NCP proteins of SARS-CoV-2 in individuals with or without SARS-CoV-2 vaccination (+vacc./−vacc.) and with or without prior COVID-19 from the DigiHero cohort. (B) Median time from positive PCR or antigen test to participation in the module for the prior COVID-19 (n = 154) and ongoing PASC groups (n = 104). All rights reserved.Ĭlinical and epidemiological parameters of the DigiHero discovery cohort and patients with PASC (A) Flow chart of the COVID-19 module of the DigiHero study. Blood profiling and single-cell data from early infection suggest the induction of these cytokines in COVID-19 lung pro-inflammatory macrophages creating a self-sustaining feedback loop.ĬOVID-19 IL-1β IL-6 PASC SARS-CoV-2 TNF cytokine long covid macrophage post-acute sequelae of COVID-19.Ĭopyright © 2022 The Author(s). PASC is not associated with autoantibodies, but with elevated IL-1β, IL-6, and TNF plasma levels, which we confirm in a validation cohort with 333 additional participants and a longer time from infection of 10 months. Of those, 67.8% report PASC, predominantly consisting of fatigue, dyspnea, and concentration deficit, which persists in 60% over the mean 8-month follow-up period and resolves independently of post-infection vaccination. We report the first 318 participants, the majority thereof after mild infections. Using a digital epidemiology approach, we recruited 8,077 individuals to the cohort study for digital health research in Germany (DigiHero) to respond to a basic questionnaire followed by a PASC-focused survey and blood sampling. Electronic address: sequelae of COVID-19 (PASC) is emerging as global problem with unknown molecular drivers.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
